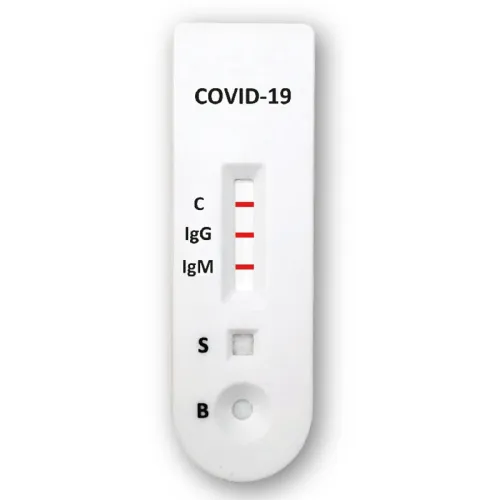
Accu-Tell® COVID-19 IgG/IgM Rapid Test Cassette (Whole Blood/ Serum/ Plasma)
Product Description
STATEMENT:
1. Accu-Tell® COVID-19 IgG/IgM Rapid Test is for professional use. It is NOT for self-testing or home use;
2. Any distributor shall notify to your local authority about the product and get the appropriate approval before you import and place the product in your local market;
3. We are not marketing, selling, or distributing Accu-Tell® COVID-19 IgG/IgM Rapid Test without FDA clearance in USA.
CATALOG
| Product Name | Specimen | Catalog No. | Quantity per box | Certificate |
| COVID-19 Rapid Test Cassette | Whole Blood/Serum/Plasma | ABT-IDT-B352 | 50T/25T | CE |
KEY POINTS
Detection for: Qualitative detection of IgG and IgM antibodies to SARS-CoV-2 in human whole blood, serum, or plasma as an aid in the diagnosis of primary and secondary COVID-19 infections.
Reading time: At 10 minutes
Positive agreement: 93.5%;
Negative agreement: 97.5%;
Total agreement: 95.5%.
INTENDED USE
ACCU-TELL® COVID-19 IgG/IgM Cassette(Whole Blood/ Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of IgG and IgM antibodies to SARS-CoV-2 in human whole blood, serum, or plasma as an aid in the diagnosis of primary and secondary COVID-19 infections.
TEST PROCEDURE
Allow the test cassette, specimen, buffer, and/or controls to reach room temperature (15-30°C) prior to testing.
1. Bring the pouch to room temperature before opening. Remove the test cassette from the sealed pouch and use it within one hour.
2. Place the test cassette on a clean and level surface.
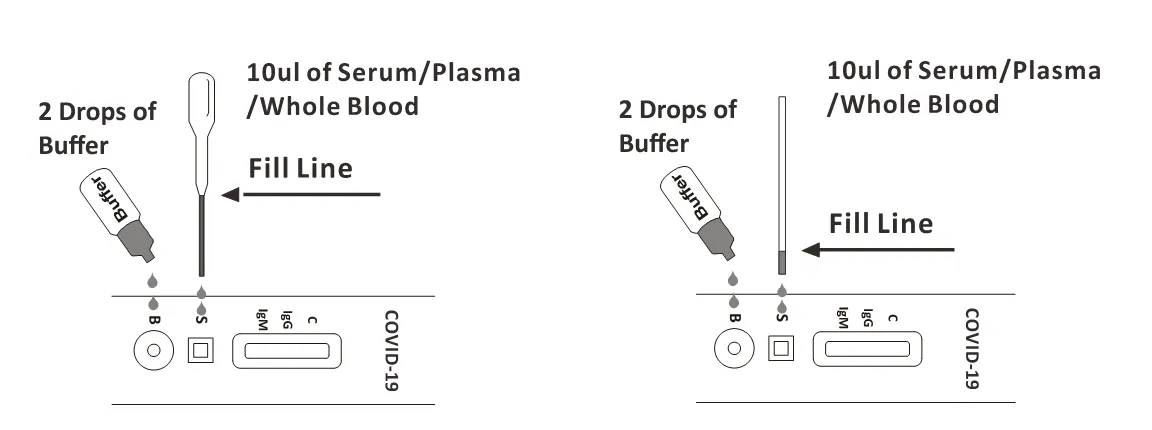 To use a dropper: Hold the dropper vertically, draw the specimen up to the Fill Line (approximately 10μl), and transfer the specimen to the specimen well (S) of the test cassette, then add 2 drops of buffer (approximately 80μl) to the buffer well (B) and start the timer. Avoid trapping air bubbles in the specimen well.
To use a dropper: Hold the dropper vertically, draw the specimen up to the Fill Line (approximately 10μl), and transfer the specimen to the specimen well (S) of the test cassette, then add 2 drops of buffer (approximately 80μl) to the buffer well (B) and start the timer. Avoid trapping air bubbles in the specimen well.
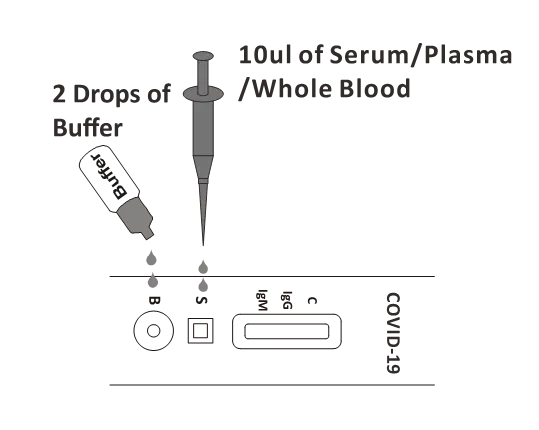
To use a micropipette (recommended): Pipette and dispense 10µl of specimen to the specimen well (S) of the test cassette, then add 2 drops of buffer (approximately 80µl) to the buffer well (B) and start the timer.
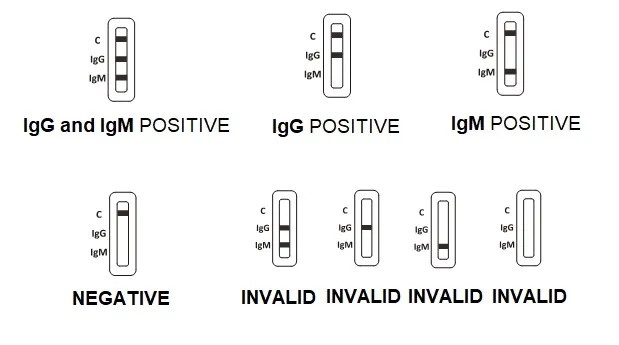
3. Wait for the colored line(s) to appear. The test result should be read at 10 minutes. Do not interpret the result after 20 minutes.
STORAGE AND STABILITY
The kit can be stored at room temperature or refrigerated (2-30°C). The test cassette is stable through the expiration date printed on the sealed pouch. The test cassette must remain in the sealed pouch until use. DO NOT FREEZE. Do not use beyond the expiration date.
PERFORMANCE CHARACTERISTICS
Positive Agreement
Positive agreement of ACCU-TELL® COVID-19 IgG/IgM Cassette (Whole Blood/Serum/Plasma) was evaluated using clinical samples collected from symptomatic subjects. All subjects were confirmed positive for COVID-19 by Nucleic Acid Test (RT-PCR) or clinical diagnosis.
Negative Agreement
Negative agreement of ACCU-TELL® COVID-19 IgG/IgM Cassette (Whole Blood/Serum/Plasma) was evaluated using clinical samples collected from symptomatic subjects. Samples were collected during the 2020 COVID-19 pandemic and all were excluded for COVID-19 by Nucleic Acid Test (RT-PCR) or clinical diagnosis.
The positive and/or negative population consisted of the following subjects.
The results show that the positive agreement of ACCU-TELL® COVID-19 IgG/IgM Cassette (Whole Blood/Serum/Plasma) is 93.5%, the negative agreement is 97.5% and the total agreement is 95.5%.
• Living in site A during the 2020 COVID-19 pandemic.
• Living in site B during the 2020 COVID-19 pandemic.
| Method | PCR Positive (Confirmed Cases) | PCR Negative(Excluded Cases) | Total | |
| Site A | IgG+ and IgM+ | 84 | 1 | 85 |
| IgM+ and IgG- | 9 | 3 | 12 | |
| IgM- and IgG+ | 2 | 0 | 2 | |
| IgM- and IgG- | 5 | 146 | 151 | |
| Site B | IgG+ and IgM+ | 58 | 0 | 58 |
| IgM+ and IgG- | 5 | 0 | 5 | |
| IgM- and IgG+ | 1 | 0 | 1 | |
| IgM- and IgG- | 6 | 10 | 16 | |
| Total | 170 | 160 | 330 | |
| Agreement | (95+64)/(100+70)=93.5%(95%CI:88.7%-96.7% ) | (146+10)/(150+10)=97.5%(95%CI:93.7%-99.3% ) | (95+64+146+10)/( 100+70+150+10)=95.5%(95%CI:93.7%-99.3% ) | |
CI means confidence interval
Cross-reactivity
ACCU-TELL® COVID-19 IgG/IgM Cassette (Whole Blood/Serum/Plasma) has been tested for anti-influenza A virus, anti-influenza B virus, anti-RSV, anti-Adenovirus, HBsAg, anti-Syphilis, anti-H. Pylori, anti-HIV, anti-HCV, anti-haemophilus influenza, ANA, H1N1, H3N2, H7N9, coronavirus HKU1, NL63, OC43, 229E and HAMA positive specimens. The results showed no cross-reactivity. Some cross reactivity was observed with samples positive for SARS-CoV antibody and Rheumatoid Factor. It is possible to cross-react with samples positive for MERS-CoV antibody.
Interfering Substances
The following potentially interfering substances were added to COVID-19 negative and spiked positive specimens.
| Analytes | Concentration | Result | |||
| Negative Specimen | Spiked with Positive Specimen | ||||
| IgG line | IgM line | IgG line | IgM line | ||
| Acetaminophen | 20 mg/dL | Negative | Negative | Positive | Positive |
| Caffeine | 20 mg/dL | Negative | Negative | Positive | Positive |
| Albumin | 2 g/dL | Negative | Negative | Positive | Positive |
| Acetylsalicylic Acid | 20 mg/dL | Negative | Negative | Positive | Positive |
| Gentisic Acid | 20 mg/dL | Negative | Negative | Positive | Positive |
| Ethanol | 1% | Negative | Negative | Positive | Positive |
| Ascorbic Acid | 2g/dL | Negative | Negative | Positive | Positive |
| Creatine | 200mg/dl | Negative | Negative | Positive | Positive |
| Bilirubin | 1g/dL | Negative | Negative | Positive | Positive |
| Hemoglobin | 1000mg/dl | Negative | Negative | Positive | Positive |
| Oxalic Acid | 60mg/dL | Negative | Negative | Positive | Positive |
| Uric acid | 20mg/ml | Negative | Negative | Positive | Positive |
None of the substances at the concentration tested interfered in the assay.
LIMITATIONS
1. ACCU-TELL® COVID-19 IgG/IgM Cassette (Whole Blood/Serum/Plasma) is for in vitro diagnostic use only. The test should be used for the detection of SARS-COV-2 antibodies in whole blood, serum or plasma specimens only. Neither the quantitative value nor the rate of increase in SARS-COV-2 antibody concentration can be determined by this qualitative test.
2. ACCU-TELL® COVID-19 IgG/IgM Cassette (Whole Blood/Serum/Plasma) will only indicate the presence of SARS-COV-2 antibodies in the specimen and should not be used as the sole criteria for the diagnosis of SARS-COV-2.
3. In the early onset of fever symptom, anti-SARS-COV-2 IgM concentrations may be below detectable levels.
4. The continued presence or absence of antibodies cannot be used to determine the success or failure of therapy.
5. Results from immunosuppressed patients should be interpreted with caution.
6. As with all diagnostic tests, all results must be interpreted together with other clinical information available to the physician.
7. Negative results do not rule out SARS-CoV-2 infection, particularly in those who have been in contact with the virus. Follow-up testing with a molecular diagnostic should be considered to rule out infection in these individuals.
8. Positive results may be due to past or present infection with non-SARS-CoV-2 coronavirus strains, such as coronavirus HKU1, NL63, OC43, or 229E.
9. This test has not been reviewed by the FDA.
10. Results from antibody testing should not be used as the sole basis to diagnose or exclude SARS-CoV-2 infection or to inform infection status.
11. Not for the screening of donated blood.
12. There may be false positive risk with the plasma in EDTA tube after a period storage.
Note: The above information is for reference use only. Please refer to the product insert provided with the products before use.
Introduction Video:
Components of ACCU-TELL® COVID 19 Rapid Test Kit:
Test Procedure of ACCU-TELL® COVID 19 Rapid Test:
Test Results of ACCU-TELL® COVID 19 Rapid Test:
View More