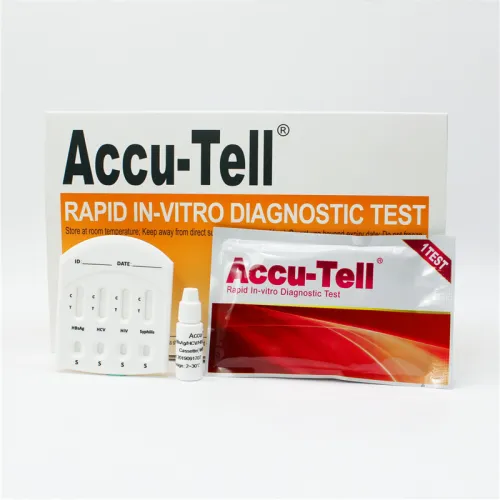
Accu-Tell® HBsAg/HCV/HIV/Syphilis Combo Rapid Test Cassette (Whole Blood/Serum/Plasma)
Product Description
CATALOG
Product Name | Specimen | Catalog No. | Quantity per box | Certificate |
HBsAg/HCV/HIV/Syphilis Combo Cassette | Whole Blood/Serum/Plasma | ABT-IDT-B235 | 15T |
KEY POINTS
Detection for: Hepatitis B surface antigen (HBsAg), antibodies to Hepatitis C Virus, antibodies to HIV type 1, type 2 and syphilis antibodies (IgG and IgM) to Treponema Pallidum (TP) in whole blood, serum or plasma.
Specimen volume: Serum or Plasma Specimen: 50μL
Whole Blood Specimens:75μL
Reading time: 10 minutes
HBsAg
Relative Sensitivity: >99.9% (97.5%CI*: 98.2%-100%)
Relative Specificity: 99.0% (95%CI*: 97.2%-99.8%)
Accuracy:99.4 %(95%CI*:98.3%-99.9%)
HCV
Relative sensitivity: 99.1% (95%CI*: 94.9%~100.0%);
Relative specificity: 99.5% (95%CI*: 98.6%~99.9%);
Accuracy: 99.4% (95%CI*:98.6%~99.8%).
HIV 1.2
Relative sensitivity: >99.99% (97.5%CI*: 97.20%~100.0%);
Relative specificity: 99.88% (95%CI*: 99.57%~99.99%);
Accuracy: 99.89% (95%CI*: 99.60%~99.99%).
Syphilis
Relative sensitivity: >99.9% (95%CI*: 97.7%~100.0%);
Relative specificity: 99.7% (95%CI*: 98.2%~100.0%);
Accuracy: 99.8% (95%CI*:98.2%~100.0%).
INTRODUCTION
1. The HBsAg Rapid Test (Whole Blood/Serum/Plasma) is a rapid test to qualitatively detect the presence of HBsAg in whole blood, serum or plasma specimen. The test utilizes a combination of monoclonal and polyclonal antibodies to selectively detect elevated levels of HBsAg in whole blood, serum or plasma.
2. The HCV Rapid Test (Whole Blood/Serum/Plasma) is a rapid test to qualitatively detect the presence of antibody to HCV in whole blood, serum or plasma specimen. The test utilizes colloid gold conjugate and recombinant HCV proteins to selectively detect antibody to HCV in whole blood, serum or plasma. The recombinant HCV proteins used in the test kit are encoded by the genes for both structural (nucleocapsid) and non-structural proteins.
3. The HIV 1.2 Rapid Test (Whole Blood/Serum/Plasma) is a rapid test to qualitatively detect the presence of antibody to HIV 1 and/or HIV 2 in whole blood, serum or plasma specimen. The test utilizes latex conjugate and multiple recombinant HIV proteins to selectively detect antibodies to the HIV 1.2 in whole blood, serum or plasma.
4. The Syphilis Rapid Test (Whole Blood/Serum/Plasma) utilizes a double antigen combination of a Syphilis antigen coated particle and Syphilis antigen immobilized on membrane to detect TP antibodies (IgG and IgM) qualitatively and selectively in whole blood, serum or plasma.
INTENDED USE
ACCU-TELL® HBsAg/HCV/HIV/Syphilis Combo Rapid Test Cassette (Whole Blood/Serum/Plasma) is a rapid chromatographic immunoassay for the qualitative detection of Hepatitis B surface antigen(HBsAg),antibodies to Hepatitis C Virus, antibodies to HIV type 1, type 2 and syphilis antibodies (IgG and IgM) to Treponema Pallidum (TP) in whole blood,serum or plasma.
TEST PROCEDURE

1. Bring the pouch to room temperature before opening it. Remove the test cassette from the sealed pouch and use it as soon as possible. Best results will be obtained if the assay is performed within one hour.
2. Place the test cassette on a clean and level surface.
For Serum/Plasma Specimens:
Hold the dropper vertically and transfer 2 drops of serum or plasma (approximately 50μL) to the specimen area, then add 1 drop of buffer (approximately 40μL), respectively. Start the timer. See the illustration below.
For Whole Blood Specimens:
Hold the dropper vertically and transfer 3 drops of whole blood (approximately 75μL) to the specimen area, then add 2 drops of buffer (approximately 80μL), respectively. Start the timer. See the illustration below.
3. Wait for the colored line(s) to appear. The test result should be read at 10 minutes. Do not interpret the result after 20 minutes.
PERFORMANCE CHARACTERISTICS
Sensitivity and Specificity
HBsAg
The HBsAg Rapid Test (Whole Blood/Serum/Plasma) has been tested with a sensitivity panel ranging from 0 to 300ng/ml. All 10 HBsAg subtypes produced positive results on The HBsAg Rapid Test (Whole Blood/Serum/Plasma). The test can detect 1 PEI ng/ml of HBsAg in whole blood/serum/plasma.
Antibodies used for the HBsAg Rapid Test (Whole Blood/Serum/Plasma) were developed against whole Hepatitis B antigen isolated from Hepatitis B virus. Specificity of the HBsAg Rapid Test (Whole Blood/Serum/Plasma) was also tested with laboratory strains of Hepatitis A and Hepatitis C. They all yielded negative results.
Method | EIA | Total Results | ||
HBsAg Rapid Test | Results | Positive | Negative | |
Positive | 205 | 3 | 208 | |
Negative | 0 | 310 | 310 | |
Total Results | 205 | 313 | 518 | |
Relative Sensitivity: >99.9% (97.5%CI*: 98.2%-100%)
Relative Specificity: 99.0% (95%CI*: 97.2%-99.8%)
Accuracy: 99.4% (95%CI*: 98.3%-99.9%)
*Confidence Intervals
HCV
The recombinant antigen used for the HCV Rapid Test (Whole Blood/Serum/Plasma) is encoded by genes for both structural (nucleocapsid) and non-structural proteins. The HCV Rapid Test (Whole Blood/Serum/Plasma) has passed a seroconversion panel and compared with a leading commercial HCV EIA test using clinical specimens.
The results show that the relative sensitivity of the HCV Rapid Test Cassette (Whole Blood/Serum/Plasma) is 99.1%, and the relative specificity is 99.5%.
Method | EIA | Total Results | ||
HCV Rapid Test | Results | Positive | Negative | |
Positive | 107 | 3 | 110 | |
Negative | 1 | 599 | 600 | |
Total Result | 108 | 602 | 710 | |
Relative sensitivity: 99.1% (95%CI*: 94.9%~100.0%);
Relative specificity: 99.5% (95%CI*: 98.6%~99.9%);
Accuracy: 99.4% (95%CI*: 98.6%~99.8%).
*Confidence Intervals
HIV 1.2
The HIV 1.2 Rapid Test (Whole Blood/Serum/Plasma) has correctly identified specimens of a seroconversion panel and has been compared to a leading commercial ELISA HIV test using clinical specimens. The results show that the relative sensitivity of the HIV 1.2 Rapid Test (Whole Blood/Serum/Plasma) is >99.99% and the relative specificity is 99.88%.
Method | ELISA | Total Results | ||
HIV 1.2 Rapid Test | Results | Positive | Negative | |
Positive | 130 | 2 | 132 | |
Negative | 0 | 1683 | 1683 | |
Total Result | 130 | 1685 | 1815 | |
Relative sensitivity: >99.99% (97.5%CI*: 97.20%~100.0%);
Relative specificity: 99.88% (95%CI*: 99.57%~99.99%);
Accuracy: 99.89% (95%CI*: 99.60%~99.99%).
*Confidence Intervals
Syphilis
The Syphilis Rapid Test (Whole Blood/Serum/Plasma) has correctly identified specimens of a seroconversion panel and has been compared to a leading commercial TPPA Syphilis test using clinical specimens. The results show that the relative sensitivity of the Syphilis Rapid Test (Whole Blood/Serum/Plasma) is >99.9% and the relative specificity is 99.7%.
Method | TPPA | Total Results | ||
Syphilis Rapid Test | Results | Positive | Negative | |
Positive | 130 | 1 | 131 | |
Negative | 0 | 299 | 299 | |
Total Result | 130 | 300 | 430 | |
Relative sensitivity: >99.9% (95%CI*: 97.7%~100.0%);
Relative specificity: 99.7% (95%CI*: 98.2%~100.0%);
Accuracy: 99.8% (95%CI*: 98.2%~100.0%).
*Confidence Intervals
Precision
Intra-Assay
Within-run precision has been determined by using 20 replicates of four different specimens containing different concentrations of HBsAg, HCV antibody, HIV 1.2 antibody and syphilis antibody. The negative, positive values were correctly identified 100% of the time.
Inter-Assay
Between-run precision has been determined by 20 independent assays on the same four different specimens containing different concentrations of HBsAg, HCV antibody, HIV 1.2 antibody and syphilis antibody. three specimens: a negative, a HCV low titer positive and a HCV high titer positive. Three different lots of ACCU-TELL® HBsAg/HCV/HIV/Syphilis Combo Rapid Test Cassette (Whole Blood/Serum/Plasma) have been tested over a 3-month period using above negative and positive specimens. The specimens were correctly identified 100% of the time.
Cross-reactivity
The HBsAg Rapid Test (Whole Blood/Serum/Plasma) has been tested by HAMA, Rheumatoid factor (RF), HAV, Syphilis, HIV, H. Pylori, MONO, CMV, Rubella, HCV, HEV and TOXO positive specimens. The results showed no cross-reactivity.
The HCV Rapid Test (Whole Blood/Serum/Plasma) has been tested by HAMA, RF, HBsAg, HBsAb, HBeAg, HBeAb, HBcAb, Syphilis, HIV, H. Pylori, MONO, CMV, Rubella and TOXO positive specimens. The results showed no cross-reactivity.
The HIV 1.2 Rapid Test (Whole Blood/Serum/Plasma) has been tested by HAMA, RF, HBsAg, HBsAb, HBeAg, HBeAb, HBcAb, HCV, Syphilis, H. Pylori, MONO, CMV, Rubella and TOXO positive specimens. The results showed no cross-reactivity.
The Syphilis Rapid Test (Whole Blood/Serum/Plasma) has been tested by HAMA, RF, HBsAg, HBsAb, HBeAg, HBeAb, HBcAb, HCV, HIV, H. Pylori, MONO, CMV, Rubella and TOXO positive specimens. The results showed no cross-reactivity.
Interfering Substances
The following potentially interfering substances were added to HBsAg, HCV antibody, HIV 1.2 antibody and syphilis antibody negative and positive specimens.
Acetaminophen: | 20 mg/dL | Caffeine: | 20 mg/dL |
Acetylsalicylic Acid: | 20 mg/dL | Gentisic Acid: | 20 mg/dL |
Ascorbic Acid: | 2g/dL | Albumin: | 2 g/dL |
Creatin: | 200 mg/dL | Hemoglobin: | 1000mg/dL |
Bilirubin: | 1g/dL | Oxalic Acid: | 60mg/dL |
None of the substances at the concentration tested interfered in the assay.
LIMITATIONS
1. This test is for in vitro diagnostic use only.
2. This test has been developed for testing whole blood/serum/plasma specimens only. The performance of the test using other specimens has not been substantiated.
3. This test is a qualitative screening assay. It is not designed to determine the quantitative concentration of HBsAg, HCV antibody, HIV 1.2 antibody or syphilis antibody.
4. The HBsAg Rapid Test cannot detect less than 1 PEI ng/ml of HBsAg in specimens.
5. As with all diagnostic tests, all results must be considered with other clinical information available to the physician.
6. If the test result is negative and clinical symptoms persist, additional follow-up testing using other clinical methods is recommended. A negative result at any time does not preclude the possibility of HBsAg and/or Hepatitis C Virus and/or HIV1.2 and/or syphilis infection.
Note: The above information is for reference use only. Please refer to the product insert provided with the products before use.
View More